
Global Adoption Status of eCTD v4.0 Across Regions
13 april 2026

The electronic Common Technical Document (eCTD) has played a pivotal role in advancing the digitalization and standardization of regulatory submissions. With the introduction of eCTD v4.0, the clinical development industry is transitioning toward a more structured, data-driven model designed to enhance efficiency through improved document reuse, greater data granularity, and more robust lifecycle management.
However, the global adoption of eCTD v4.0 remains uneven. Regulatory authorities across key regions, including the United States, Europe, and Japan, are progressing at varying paces, each following distinct timelines, implementation strategies, and levels of preparedness.
Global Overview of eCTD v4.0 Adoption
Although the standard was finalized by the International Council for Harmonization (ICH), its implementation has been gradual. Adoption is currently in a hybrid phase, where:
- eCTD v3.2.2 remains the dominant and fully operational standard worldwide
- eCTD v4.0 is being introduced through pilot programs, voluntary submissions, or phased implementations
- Mandatory timelines vary significantly by region
The period from 2024 to 2026 represents a critical transition phase, with major agencies beginning active adoption programs.
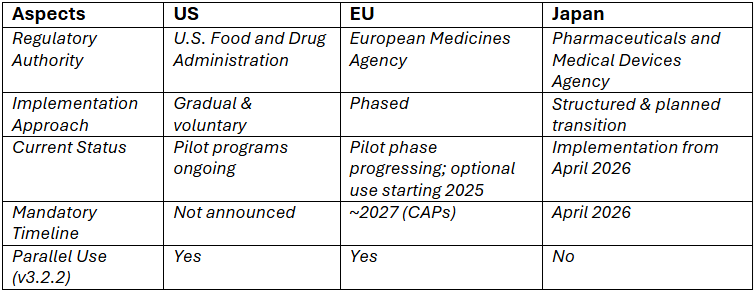
United States: Gradual and Voluntary Transition
The FDA has taken a phased and cautious approach toward eCTD v4.0 adoption.
- Since September 16, 2024, the FDA has begun accepting voluntary eCTD v4.0 submissions for new applications
- eCTD v3.2.2 continues to be the primary operational standard
- No official mandatory deadline has been announced for the transition to eCTD v4.0
The strategy focuses on industry readiness and controlled implementation, allowing sponsors to transition at their own pace while continuing to use eCTD v3.2.2 in parallel.
Europe: Phased Implementation Strategy
The EMA is initiating a phased and voluntary transition to eCTD v4.0 for Centrally Authorized Products (CAPs) starting in 2025, with full implementation anticipated around 2027.
This strategy emphasizes early technical piloting in collaboration with software vendors and Marketing Authorization Holders (MAHs) to ensure system readiness and smooth adoption. eCTD v3.2.2 remains accepted during the transition period.
EMA’s strategy emphasizes controlled implementation, stakeholder engagement, and validation readiness before full enforcement.
Japan: Leading the Global Transition
Japan, through the Pharmaceuticals and Medical Devices Agency (PMDA), is at the forefront of eCTD v4.0 adoption:
- Pilot programs and preparatory activities have been completed
- Mandatory implementation is expected from April 2026
Japan’s proactive stance positions themselves as the first major regulatory market to fully implement eCTD v4.0, setting a benchmark for other regions.
Other Regions: Emerging Adoption Trends
Beyond the major markets, several regulatory authorities are preparing for eCTD v4.0:
- Canada and Australia: Planning voluntary adoption around 2026, with mandatory timelines anticipated later
- Brazil (ANVISA) and other emerging markets: Engaged in pilot programs or exploratory phases, with some regions still awaiting clearer implementation roadmaps
eCTD v4.0 Adoption Comparisons: US vs EU vs Japan
Key Challenges Impacting Global Adoption
Despite clear benefits, several challenges are influencing the pace of adoption:
- Technical complexity of transitioning from document-based to data-driven submissions
- Training and upskilling needs for regulatory and publishing teams
- Lack of synchronized global timelines, leading to prolonged hybrid submission environments
As a result, organizations must manage parallel processes for eCTD v3.2.2 and v4.0, increasing operational complexity.
How PLG Enables a Seamless Transition to eCTD v4.0
At ProductLife Group (PLG), we support regulatory submissions across all major global regions and continuously align ourselves with evolving industry developments. Through active participation in eCTD v4.0 pilot programs, we proactively strengthen our readiness to meet emerging regulatory requirements.
PLG has developed robust capabilities to efficiently manage both eCTD v3.2.2 and v4.0 submissions. This enables us to support clients in diverse regulatory environments worldwide, ensuring high-quality deliverables aligned with regional regulatory expectations.
We ensure that our clients remain compliant, efficient, and well-prepared for the ongoing global shift toward structured, data-driven regulatory submissions.
Looking for support? Contact our team today:
Register to our news and events
Go to our Events to register
Go to our News to get insights
