
Resilient Pharmaceutical Supply Chains in Times of Global and Geopolitical Crisis: Comparative Strategies Across Australia, France, Italy, Spain, and the UK
10 march 2026

An expert analysis of pharmaceutical supply chain resilience during crises, focusing on national strategies, regulatory measures, and industrial interventions across Europe and Australia.
Structural Vulnerabilities in Global Pharmaceutical Supply Chains
Recent years have highlighted the fragility of global pharmaceutical supply chains. Multiple converging crises (including the COVID-19 pandemic, geopolitical conflicts, rising energy costs, and climate-related disruptions) have revealed systemic weaknesses in the production, distribution, and availability of medicines. These disruptions have been particularly pronounced in essential therapeutic areas, leading to persistent drug shortages with significant implications for patient care and healthcare systems.
From a patient perspective, shortages often result in treatment delays, therapy interruptions, and adverse health outcomes, while also causing uncertainty and anxiety related to changes in medication regimens. Healthcare professionals experience considerable operational strain: studies estimate that up to seven hours per week can be devoted to sourcing alternatives or liaising with suppliers, limiting time for direct patient care and increasing costs for health systems.
At the European level, the European Medicines Agency (EMA) plays a critical role in monitoring and mitigating medicine shortages. EMA coordinates with national competent authorities to enhance early detection of supply disruptions, implement digital tracking systems for critical medicines, and promote continuity planning with marketing authorization holders. Complementing these efforts, the EU Pharma Package, adopted in 2023 and entering into force in 2026, strengthens the EU’s regulatory framework to address shortages systematically. The pharma package proposal aims to strengthen these types of control on the supply of medicines by providing a new obligation that MAHs ensure sufficient supply of medicines within individual EU member states. It is unclear exactly how this measure will be promulgated or enforced, but it will provide an extra element of uncertainty for those aiming to market products and in considering where they choose to do so, given the global markets in which medicines are manufactured and supplied. It will include penalties for those companies that fail to comply with the new regulation.
Against this backdrop, it is increasingly important to understand how national strategies can enhance supply chain resilience. This analysis examines the approaches adopted by Australia, France, Italy, Spain, and the United Kingdom, focusing on prevention of shortages, strengthening systemic resilience, and ensuring equitable access to essential medicines. Emphasis is placed on the integration of digital infrastructure, regulatory agility, public-private collaboration, and domestic manufacturing capabilities to achieve long-term sustainability.
Comparative Policy Analysis Across Jurisdictions
A qualitative comparative methodology was employed to evaluate national responses to medicine shortages. Data sources included publicly available documentation from national medicines agencies, health ministries, and parliamentary reports, alongside digital monitoring tools and shortage databases. The study also considered EU-level regulatory developments, particularly Regulation (EU) 2022/123, and initiatives such as EU FAB, HERA, and the Critical Medicines Alliance, to understand alignment or divergence in national strategies.
Interventions were assessed for potential impact on shortage mitigation (1= low impact; 3= max impact), with attention to proactive measures such as early reporting requirements, digital traceability systems, and continuity planning. By adopting a multidimensional approach, the study identifies patterns, gaps, and innovative practices that could inform cross-border regulatory and industrial strategies.
National Strategies and Policy Measures
Australia
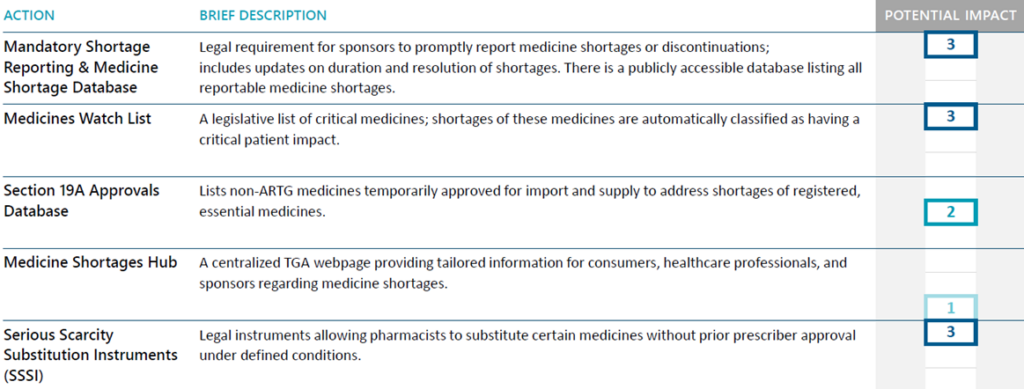
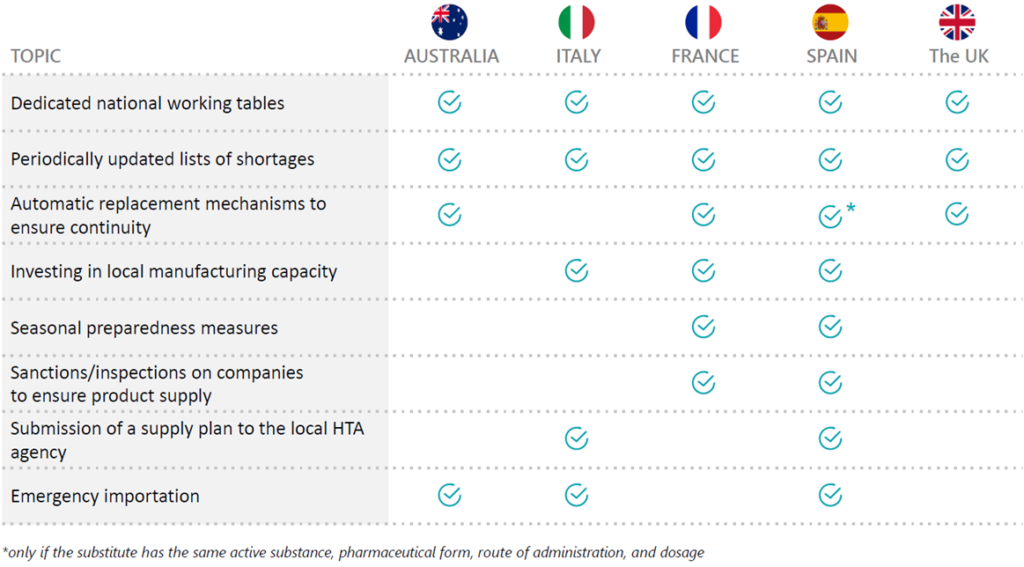
In Australia, the Therapeutic Goods Administration (TGA) defines a medicine shortage as any scenario where supply is unlikely to meet expected demand within the next six months. The Australian strategy emphasizes early detection, mandatory reporting, and structured communication with healthcare professionals. Sponsors are legally required to notify the TGA promptly of anticipated shortages or discontinuations, which are cataloged in a publicly accessible database. Critical medicines are monitored via the Medicines Watch List, which automatically flags shortages with high patient impact. Pharmacists are empowered through the Serious Scarcity Substitution Instruments (SSSI) to provide certain alternatives without prior prescriber approval, ensuring continuity of care while mitigating disruption risk.
Italy
Italy’s approach, coordinated by the Italian Medicines Agency (AIFA), focuses on proactive prevention and strengthening domestic production of active pharmaceutical ingredients (APIs) and excipients. Central to Italy’s strategy is the integration of dispensing, consumption, and stock data into a system that informs timely interventions. The Permanent Technical Committee, comprising regional health authorities, pharmacies, and general practitioners, monitors shortages and coordinates remedial measures. Companies are required to submit self-declarations confirming capacity to maintain uninterrupted supply during the Health Technology Assessment (HTA) process, reflecting a preventive regulatory stance aimed at reducing systemic vulnerabilities.

France
France has implemented a comprehensive set of measures to enhance supply chain resilience and encourage local production. Manufacturers are mandated to maintain 2-4 months of safety stock for medicines of major therapeutic interest (MITMs), enforced through inspections and financial penalties. The National Roadmap 2024–2027 outlines a multi-ministerial strategy encompassing real-time monitoring, therapeutic substitution, rapid diagnostic testing, reshoring initiatives, and digital platforms for supply chain transparency. Industrial policies under France Relance and France 2030 support over 100 pharmaceutical projects to increase domestic API and essential drug production. Seasonal and emergency contingency plans further ensure responsiveness to demand peaks or crisis situations.

Spain
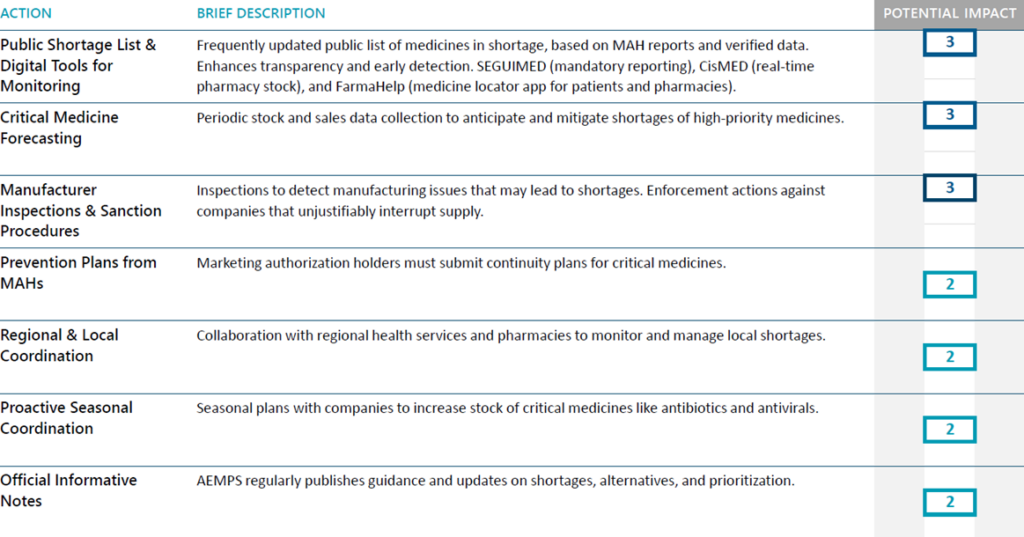
Spain has adopted a multi-layered approach combining regulatory oversight, industrial policy, and digital monitoring. The Agencia Españolade Medicamentos y Productos Sanitarios (AEMPS) maintains a publicly accessible shortage list and digital tools such as SEGUIMED, CisMED, and FarmaHelp to enhance transparency and early detection. Marketing authorization holders are obliged to submit continuity plans for critical medicines, and inspections are conducted to prevent unjustified supply interruptions. Seasonal coordination ensures sufficient stock of high-priority medicines, including antibiotics and antivirals, while regional collaboration facilitates rapid response at local and national levels.
United Kingdom
The UK relies on early notification by marketing authorization holders (MAHs), digital reporting via the DaSH portal, and centralized coordination by the Department of Health and Social Care (DHSC). Mandatory early reporting ensures anticipated shortages are flagged well in advance, while Serious Shortage Protocols allow pharmacists to substitute medicines under predefined protocols. Real-time alerts via the Central Alerting System enable rapid mitigation during critical shortages, complemented by a dedicated DHSC Medicines Supply Team that liaises with MAHs to maintain supply continuity.

Converging Principles and Strategic Insights
Despite differences in national implementation, certain principles are common across jurisdictions. Mandatory early reporting of shortages, supported by real-time digital infrastructure, enhances transparency and accelerates response times. Legal frameworks allowing pharmacist-led substitution maintain continuity of care during critical shortages. Investments in local manufacturing capacity reduce dependence on international supply chains, while continuity planning and seasonal preparedness strengthen system resilience in the face of geopolitical, market, or environmental disruptions.
Integration of regulatory agility, digital monitoring, and public-private collaboration emerges as a key enabler of sustainable supply chain resilience. Countries that proactively align industrial policies with health system needs are better positioned to anticipate shortages and mitigate patient impact.
Conclusion
Pharmaceutical supply chain resilience requires a comprehensive approach that blends regulatory oversight, industrial policy, and technological innovation. Best practices include mandatory early reporting, substitution protocols for pharmacists, investment in domestic production of essential drugs and APIs, and implementation of continuity and seasonal preparedness measures. By adopting these strategies, healthcare systems can minimize patient disruption, optimize operational efficiency, and build resilience against increasingly complex global crises.
The Author
 |
Mariangela Prada, Intexo Società Benefit – A PLG Company |
 |
Tanya van der Wall, Commercial Eyes – A PLG Company |
 |
Sébastien Faure, Nextep – A PLG Company |
 |
Letizia Rossi, Intexo Società Benefit – A PLG Company |
 |
Robin Puzenat, Nextep – A PLG Company |
 |
Vincent Victory, Commercial Eyes – A PLG Company |
 |
Paloma Gonzáles, Outcomes’10 – A PLG Company |
References
- Phuong JM, et al. The impacts of medication shortages on patient outcomes: A scoping review. PLoS One. 2019;14(5):e0215837.
- EAHP. EAHP 2023 Shortage Survey Report. Link
- PGEU GPUE. Medicine Shortages – PGEU Survey 2022 Results. Link
- Council of the European Union & European Parliament. Regulation (EU) 2022/123.
- Therapeutic Goods Administration (TGA). Link
- Agenzia Italiana del Farmaco (AIFA). Link
- Agence Nationale de Sécurité du Médicament et des Produits de Santé (ANSM). Link
- Agencia Española de Medicamentos y Productos Sanitarios (AEMPS). Link
- Legislation.gov.uk. Link
Register to our news and events
Go to our Events to register
Go to our News to get insights
